Metal oxides have occupied a prominent place in heterogeneous catalysis with broad applications in ammonia synthesis, Fischer–Tropsch process, and so on. Extensive research efforts have been put forward to understand the nature of metal oxides as catalysts as well as support materials for energy conversion and storage. One of the primary missions of catalysis research is to design and optimize the activity and selectivity of catalysts at the atomic level, which calls for a comprehensive understanding of the structure-property relationship. Core-shell nanostructures with an ultrathin shell are widely used as catalysts to improve structural stability and catalytic activity in a controlled way. By rationally tuning the core/shell combination, a range of core-shell nanostructures with tailorable properties can be achieved, providing a versatile and intriguing platform to study the structure-property relationship. Over the past decade, a plethora of metal-based core/shell combinations have been realized, and their structure-property relationships are exploited for a wide range of purposes. When it comes to oxide-based core-shell nanostructures, the study is very limited, which holds an enormous potential for innovation.
We are developing an in-depth understanding of oxide core-shell nanostructures, with particular emphasis on the structure-property relationship. The obtained correlation will then be applied to design cathode materials of “independently tunable” metal oxide supports and rechargeable metal-air batteries. In the long term, the outcomes of this research program will lead to the development of a general theory for describing the surface reactivity of metal oxides.
Project (a): Subsurface engineered metal oxides as “independently tunable” supports
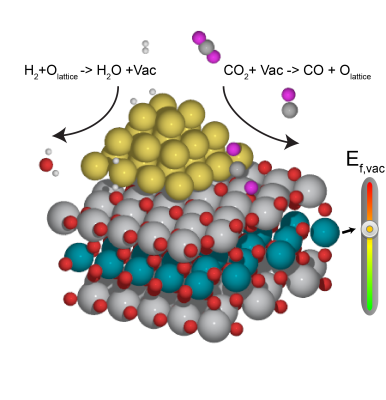
Another important role metal oxides play in the heterogeneous catalysis has been as support materials, especially in oxide-supported metal NP catalysts. It has long been recognized that the reducibility of the oxide support has a non-trivial influence on the performance of the oxide-supported metal catalyst, including electronic and morphological modifications of the metal NP, as well as the cooperation of reactions at the metal-support interface. One of the most common approaches to investigate the role of support reducibility is to immobilize metal NP on different metal oxides. However, the downside of this approach is that other parameters, such as support surface morphology and lattice misfit between support and NP, are also inevitably altered, which raises biases in comparisons. Our recent work revealed that the reducibility of the oxide support surface, often represented by O vacancy formation energy, can be tuned over a wide range by subsurface substitution, while the morphological features of the surface remain the same. Hence, we look into the development of subsurface engineered metal oxides (SEMO) as "independently tunable" supports to understand the metal-support interaction.
First, SEMO supports allow us to clarify the dependence of metal particle morphology on the support reducibility. Global optimization of a Cu8 cluster will be performed on the stoichiometric rutile TiO2(110) surfaces with subsurface Ti atoms substituted by Nb, Ru, and Ag, i.e.Cu8/[TiO2]1/[MO2]1/TiO2 (M= Nb, Ru, and Ag). An optimized genetic algorithm (GA) with focus on supported nanostructures by Hammer et. al. will be adopted for the GO. The hypothesis will then be examined by comparing the global minimum structures of Cu8 clusters on pure and SE-TiO2 supports. CO adsorption energy on different sites of Cu8 clusters will also be probed to assess the influence of particle morphology on the reactivity.
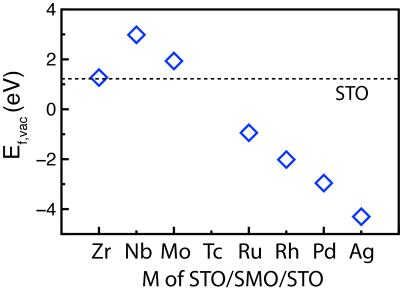
The next research activity of this project is to prove that SEMO supports can fine-tune the activity and selectivity of supported NP catalysts. The reverse water gas shift (RWGS) reaction will be studied as a probe reaction on previous optimized Cu/SE-TiO2 structures. The RWGS reaction is the thermochemical reduction of CO2 by H2 to CO and water. Figure 1 illustrates the direct C-O bond cleavage pathway on the Cu8/SE-TiO2 catalyst. H2 almost immediately dissociates on the Cu NP with a subsequent spillover of hydrogen atoms from metal to oxide supports.CO2 adsorbs most likely at the metal-oxide perimeter via a configuration with the C atom of CO2* bound to the metal site and one of the O atoms attached to the surface O vacancy. The dissociation of CO2* to CO* and O* leaves O atoms fulfilling surface O vacancies, which are later regenerated by the spillover of hydrogen atoms. Formed CO* can either desorbs as a product CO(g) or be further hydrogenated to value-added C1 products, such as CH3OH. Consequently, strong CO2adsorption at the interface benefits the overall conversion, and high CO affinity improves the selectivity towards CH3OH.35Using DFT simulations, we will compute CO2and CO adsorption energies to gauge the activity and selectivity of RWGS reaction on Cu/TiO2and Cu/SE-TiO2 (M=Nb, Ru, and Ag). Based on the Ef,vac trend of SE-STO (Figure 2), the Ru and Ag-substituted TiO2surface should get more reduced, and an improved overall conversion with a higher yield to methanol is expected. Vice versa, Nb doping will lead to the opposite trend. A comprehensive investigation of full reaction pathways combined with kinetic Monte Carlo simulations will be followed to validate the above prediction. Findings from this project will elucidate the effect of support reducibility on particle morphology, reactivity, and selectivity of supported NP catalysts. The alloy-core@shell scheme from Project 1(b) can also be employed, when a more precise tuning is needed. In the long run, application of “tunable” SEMO supports will be extended to other oxide-supported NP catalyzed applications.
Project (b): Alloy-core@shell perovskites for cathode materials of rechargeable metal-air batteries.
Rechargeable metal-air batteries have emerged as a new class of energy storage system with remarkably high energy density. However, the development and broad application of this highly innovated technology are hampered by the lack of suitable cathodic materials, which are required to efficiently catalyze both the oxygen reduction reaction (OER) and the oxygen reduction reaction (ORR). Perovskites with a general ABO3 formula hold the promise of being a new class of OER/ORR bifunctional catalysts.
Our previous work on nanoparticle(NP) catalysis advocates the development of the alloy-core@shell architecture for efficient, robust, and tunable catalysts. The alloy-core@shell architecture consists of a homogeneous shell made of noble materials and a random alloy core of tunable composition. The noble shell protects the reactive core from degradation during the catalytic processes, and the alloy-core composition allows for fine-tuning of the catalytic properties. For a metal-based system, we revealed that there is a universal linear correlation between shell reactivity and core composition. Employing this design strategy, we successfully developed PdAu@Pt NP for the ORR, which surpassed the performance of Pt NPs. Here, we propose to generalize the application of alloy-core@shell architecture to perovskite oxide catalysts.
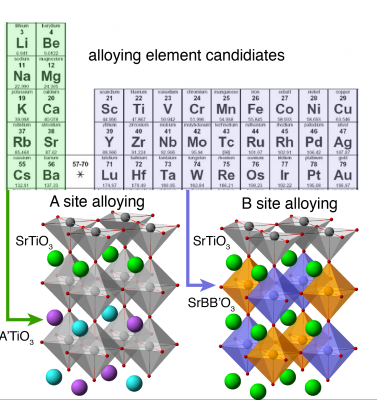
Building the correlation between core composition and surface reactivity for alloy-core@shell perovskites is a crucial task. As an initial study, we will choose SrTiO3 (STO) as the shell material, since STO is OER inert and one of the most abundant perovskites. The alloy-core is assembled by randomly substituting 2-3 unit cells (u.c) of subsurface Sr or Ti atoms with other transition metals. As illustrated in Figure 1, two strategies for the core alloying will be tested, namely A site alloying (AxA'1-x TiO3) and B site alloying (SrBxB'1-xO3). For a given alloying ratio x, O and OH adsorption energies will be calculated on a number of possible configuration(s) to get the average and the distribution of adsorption energies. Eads(x), the average adsorption energy, can thus be expressed as a function of the alloying ratio x. By comparing the correlation obtained from A site alloying with the one from B site alloying, we will be able to illuminate the dependence of surface reactivity on the core composition and identify which tuning strategy is more efficient.
With the establishment of Eads(x) for alloy-core@shell perovskites, we move on to its application as OER/ORR bifunctional catalysts. In theory, the performance of an OER/ORR bifunctional catalyst can be defined as the sum of its OER and ORR overpotential. Following the scheme developed by Nørskov et.al., we consider OH*, O* and OOH* intermediates for constructing free energy diagrams of four consecutive proton and electron transfer steps. Applying the universal linear scaling of ∆GOOH = ∆GOH + 3.2, the OER/ORR bifunctional overpotential OER/ORR can be expressed as the following:
ηOER/ORR =ηOER + ηORR
= max{∆GOH, ∆GO-∆GOH, ∆GOOH − ∆GO, 4.92 − ∆GOOH} - 1.23 + 1.23 - min{∆GOH, ∆GO-∆GOH, ∆GOOH − ∆GO, 4.92 − ∆GOOH}
= max{∆GOH, ∆GO-∆GOH, 3.2-(∆GO-∆GOH), 1.62 − ∆GOH} - min{∆GOH, ∆GO-∆GOH, 3.2-(∆GO-∆GOH), 1.62 − ∆GOH}
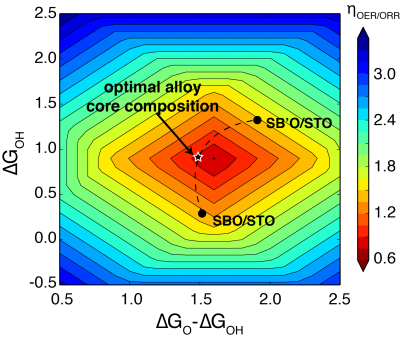
Figure 2 shows the contour plot of ηOER/ORR as a function of ∆GOH and ∆GO-∆GOH, and demonstrates the procedure for optimizing the B site alloy-core composition. Assuming that we have acquired the adsorption energies for SBO/STO and SB’O/STO as marked on the contour plot, a dashed line is drawn to connect the two points using the earlier established Eads(x). The exact shape of this line depends on the linearity of Eads(x). In a metal-based system, Eads(x) is a linear function and points are connected with straight lines. The optimal ratio is given at the point where the curvature of the line is a norm to the gradient of the 2D volcano or can be simply determined by line search along the dashed line. The same procedure is applicable to A site alloying perovskites as well.
Our method provides a general scheme to computationally design and optimize alloy-core@shell perovskites towards the top of “volcano” where conventional perovskites fail to achieve. Ultimately, we can generalize the application of alloy-core@shell architecture to other materials and applications.